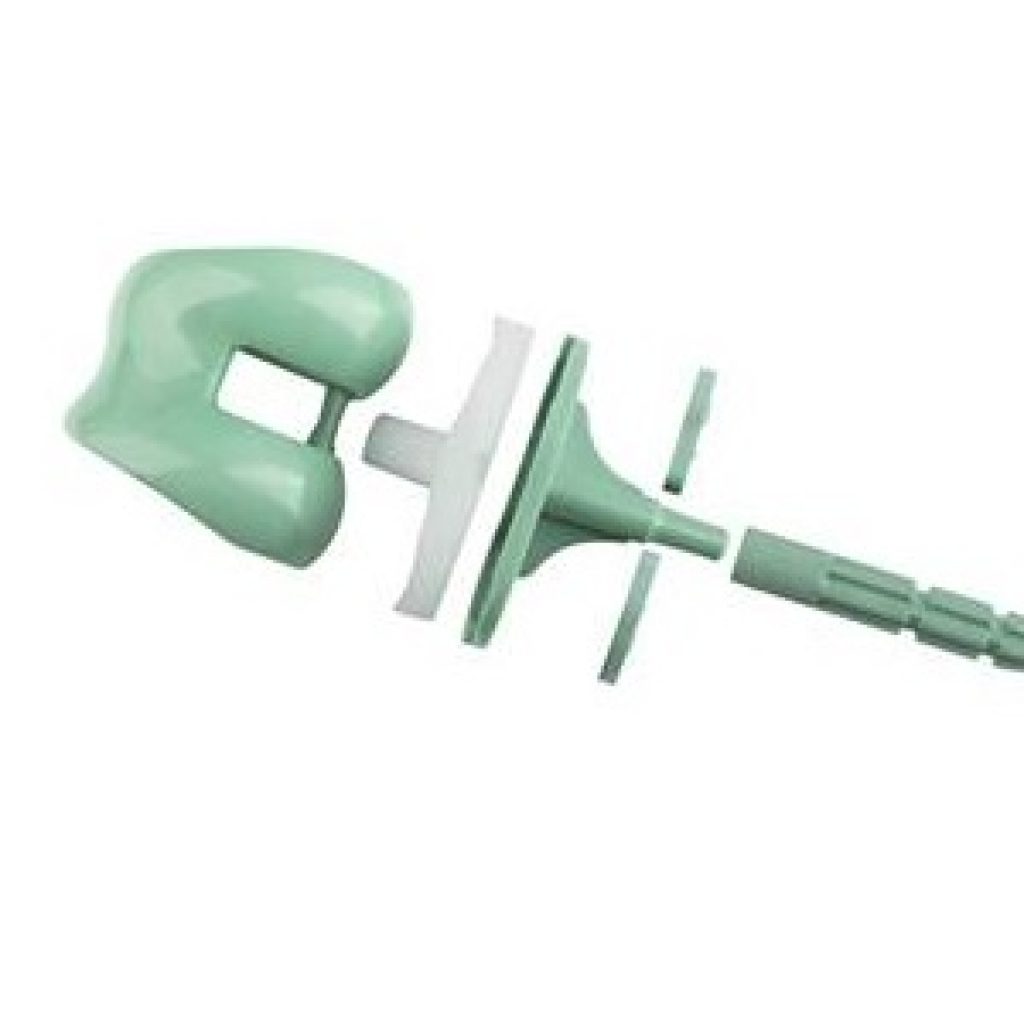
Solvay, a leading name in polymers & advanced materials industry, has announced that Okani Medical Technology has successfully developed an all polymer knee implant using its state-of-the-art Zeniva® polyetheretherketone (PEEK). Reportedly, Solvay will bring to light its new ORGknee™ implant in multiple sizes at the annual meeting of the American Academy of Orthopedic Surgeons (AAOS) 2018 that was held in New Orleans from March 10 to March 13.
According to reports, Okani’s novel ORGKnee implant offers a longer service life at a comparatively low cost than the conventional metal-based implant systems. Sources reveal that the PEEK-based knee implant showed around 50% less wear than the metallic implant as computed by material loss over 3 million cycles. Reportedly, Okani gauged Zeniva PEEK’s suitability for tibial and femoral tray components of its knee implants as per the ISO 14243-1:2009 standards, which simulates regular walking behavior for about a decade.
Solvay’s reputation as a highly innovative company in the global polymers and advanced materials industry has been the prime factor that has prompted Okani to partner with the firm, cite reliable sources. For the record, Solvay is one of the leading suppliers in polymers industry, delivering advanced & high-performance polymer materials for healthcare.
The chief technology officer at Okani, Zhonglin Zhu, in a statement said that Zeniva’s injection molding capabilities have made large-scale production of ORGKnee implants possible within a fraction of the cost & time of what it takes to manufacture metal implants. These in fact, take around three months to manufacture, machine, & polish using methods that pose risks to both workers & the environment.
It has been reported that the ORGKnee implant will enter the pre-clinical trials in April and would then undergo the standard clinical trials by the China’s FDA in September 2018. However, the commercial launch of ORGKnee solution has been scheduled for 2020 after all the CFDA approvals are completed.